|
The reason is because the PI would be in the middle and still would be considered the same structure as the lowest pka in your calculation (which is the natural form). Once you see the molecular is neutral, use that Pka and the next Pka to calculate the PI. Say the overall charge is +1, you would then use the first Pka value to get the neutral molecule which is usually by deportonating the COOH group (- charge. First, you consider the overall charge of the molecular by generally looking at how many NH3+ are there in the first place. so you can deduct that the other pka is the pkR. You should know that the pkA of all COOH is 2 and NH3 is 10 for all amino acids. You can make it easier to see by putting the pkA value between each structure from lowest to highest (you ahve 4 structures so that leave 3 spaces between them= 3 pkA value). ect.to calculate the pI, you only concern yourself with pkA associated with the -1 to 0, and the 0 to +1 change, which is what " average the two pKa values on either side of the neutral form of histidine" mean. Notice the trend, as you increase the pH, you are taking H+ away from the molecules, whichever functional group that have the lowest pkA will lose the H+ first, the highest pkA will lose it last. and then you can know that the pH have to pass though pkA of 10 for the 0 to -1 change to occur. So.that pH have to pass through which value? it is the pkR value which is 6. you can use this to find out that from +1 to 0, it is the R side chain (NH+) becoming NH, that cause the change in overall charge. The principle is simple, lets say when the overall charge is +2, as it is changing from being +2 to +1, the pH have to be higher than a certain value for the OH to become O-, that value is the pkA of COOH group, which is 2 (you can see why if you review acid-base equilibrium and definition of pka). Taht being said the picture on that website is lacking the labelling for pkA. The equation used to scale raw log(P) values to the scaled values given is as follows: Scaled Parameters = (Raw Parameters + 2.061)/4.484.Click to expand.You should know that the pkA of all COOH is 2 and NH3 is 10 for all amino acids. The hydrophobicities given are the "Scaled" values from computational log(P) determinations by the "Small Fragment Approach" (see "Development of Hydrophobicity Parameters to Analyze Proteins Which Bear Post- or Cotranslational Modifications" Black, S.D. Molecular weights for J and X were taken from Geneious. Where do amino acid hydrophobicity values come from? To calculate this, we use the same algorithm as biojava. Per ProtParam, "a protein whose instability index is smaller than 40 is predicted as stable, a value above 40 predicts that the protein may be unstable." How is a protein's instability index calculated? 
The extinction coefficient is calculated by per standard calculations using Beer's Law, via an algorithm from. How is a protein's extinction coefficient calculated? We calculate the theoretical pI by using the pKa's of amino acids and summing the net charge across the protein at a given pH, searching with our algorithm for the pH at which the net charge is zero. The pI (isoelectric point) refers to the pH at which the net charge of a protein is zero. How is a protein's isoelectric point (pI) calculated? 
Molecular weights for B and Z are non-standard and were reverse engineered from These are the average isotopic masses, as opposed to the monoisotopic mass. 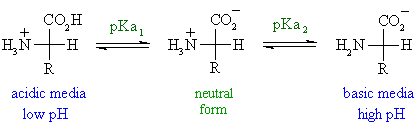
We have lightly modified constants from biopython, to match results from expasy. In Benchling, the following molecular weights are assigned to amino acids: The molecular weight of a protein is calculated by adding up the molecular weights of the amino acids, per standard calculations. How is protein molecular weight calculated?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
